- Health & Medical
Support for Healthcare Systems & Research
We provide precise translation support for healthcare and research organizations. Our focus is on regulatory compliance and accuracy across critical medical documentation.

- What It Is
Clear Messaging Supports Clinical Compliance
Medical translations must be handled with precision across languages and regulatory environments. From informed consent forms and patient materials to clinical trial documentation and device instructions, terminology requires expert oversight by linguists with healthcare experience. This structured approach helps reduce liability, support compliance, and ensure critical medical information is clearly understood.
- Why Northwest Translations
Trusted. Certified. Proven.
ISO‑Certified
ISO 17100 Certified, earning a 99.2% score on our latest quality audit and delivering consistently high-quality translations.
30+ Years of Expertise
Over 30 years of experience delivering reliable, trusted translation services across diverse industries.
No Long-Term Contracts
No long-term contracts required, giving you greater flexibility with every project.
Secure Data
SOC 2 Data Security Certified, providing the highest level of data protection for all projects.
Reliable Messaging Across Healthcare Systems
When compliance and patient safety are on the line, precision matters. Our experts support regulated medical teams with terminology accuracy across every language.
- The Process
How It Works
Submit Your Request
Leave your request using the contact form on our website or by sales@nwtranslations.com. Our manager will get in touch with you.
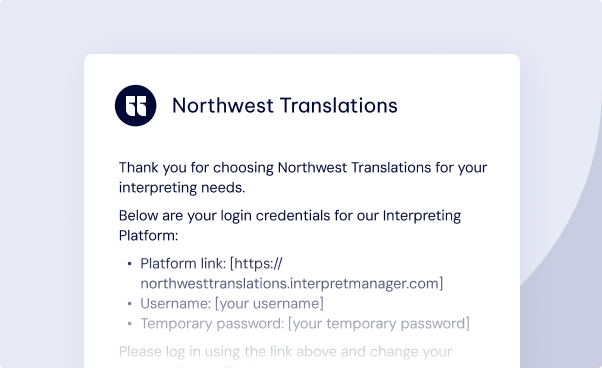
Create Your Account
You will receive an email with your credentials and link to access to our lnterpreting Platform.
Choose Your Connection Method
We offer several ways to connect: Phone (for US clients only), Web, and Mobile App (from Google Play or App Store).

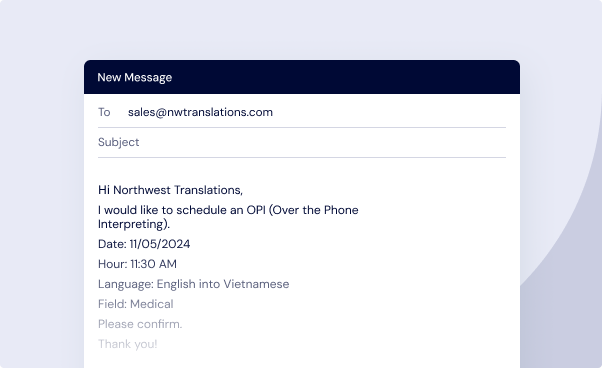
Schedule an Interpreter (If Needed)
Schedule audio or video remote interpreting by emailing sales@nwtranslations.com at least 48 hours before your meeting.
- Languages
We translate 98% of the world's languages.
We offer language support across nearly every region, helping you connect clearly with global audiences through accurate, culturally aware translations you can rely on.
- testimonials
Translating Expertise Into Results

“The translation services provided have consistently been of high quality and timely delivery. Time-sensitive requests are handled with professionalism and efficiency, which has been greatly appreciated.”


“We’ve appreciated our partnership over the last 11 years, including the responsiveness of your team and ease of collaboration. Thank you! Looking forward to many more years to come.”


“NWT consistently delivers high-quality service on time, every time.”


“The translation services provided have consistently been of high quality and timely delivery. Time-sensitive requests are handled with professionalism and efficiency, which has been greatly appreciated.”


“We’ve appreciated our partnership over the last 11 years, including the responsiveness of your team and ease of collaboration. Thank you! Looking forward to many more years to come.”


“NWT consistently delivers high-quality service on time, every time.”

- FAQ
Questions
Answered
How do you ensure accuracy in clinical trial translations?
Clinical trial documentation requires strict terminology consistency and regulatory precision. Our linguists specialize in healthcare and research environments and follow a structured review process aligned with ISO 17100 standards. Every project undergoes multi-step quality control to reduce risk and ensure accuracy across languages.
Can you translate informed consent forms (ICFs) for IRB submission?
Yes. We regularly translate informed consent forms and other patient-facing trial materials for IRB review. Our team understands the importance of clarity, cultural appropriateness, and regulatory compliance to support smooth approvals and participant understanding.
Do you support medical device documentation and labeling?
We translate instructions for use (IFUs), labeling, regulatory submissions, and technical device documentation. Our linguists work with consistent terminology databases to ensure alignment across user manuals, on-screen interfaces, and compliance materials.
How do you handle rare or hard-to-source languages in healthcare settings?
We work in over 100 languages, including rare and tribal languages often required for diverse patient populations or global trials. Our established network of vetted linguists allows us to maintain quality standards even in less common language pairs.
What is your process for urgent medical or research documents?
We understand that clinical environments operate on tight timelines. Our process ensures rapid response times and structured oversight, and we do not charge additional rush fees for time-sensitive projects.
Confidence In Every Submission
From consent forms to regulatory filings, rely on our specialists to support your healthcare systems and research teams with accurate terminology and structured quality control.